Help & Support
All Test
All Tests
- Blood Test
Popular Tests
- Plasma Test
Popular Tests
- Cancer Test
Popular Tests
- Liver Test
Popular Tests
-
- Kidney Test
Popular Tests
- Pregnancy Test
Popular Tests
- Thyroid Test
Popular Tests
- Blood Test
Popular Tests
- Plasma Test
Popular Tests
- Cancer Test
Popular Tests
- Liver Test
Popular Tests
-
- Kidney Test
Popular Tests
- Pregnancy Test
Popular Tests
- Thyroid Test
Popular Tests
Packages
Packages
0
No products in the cart.









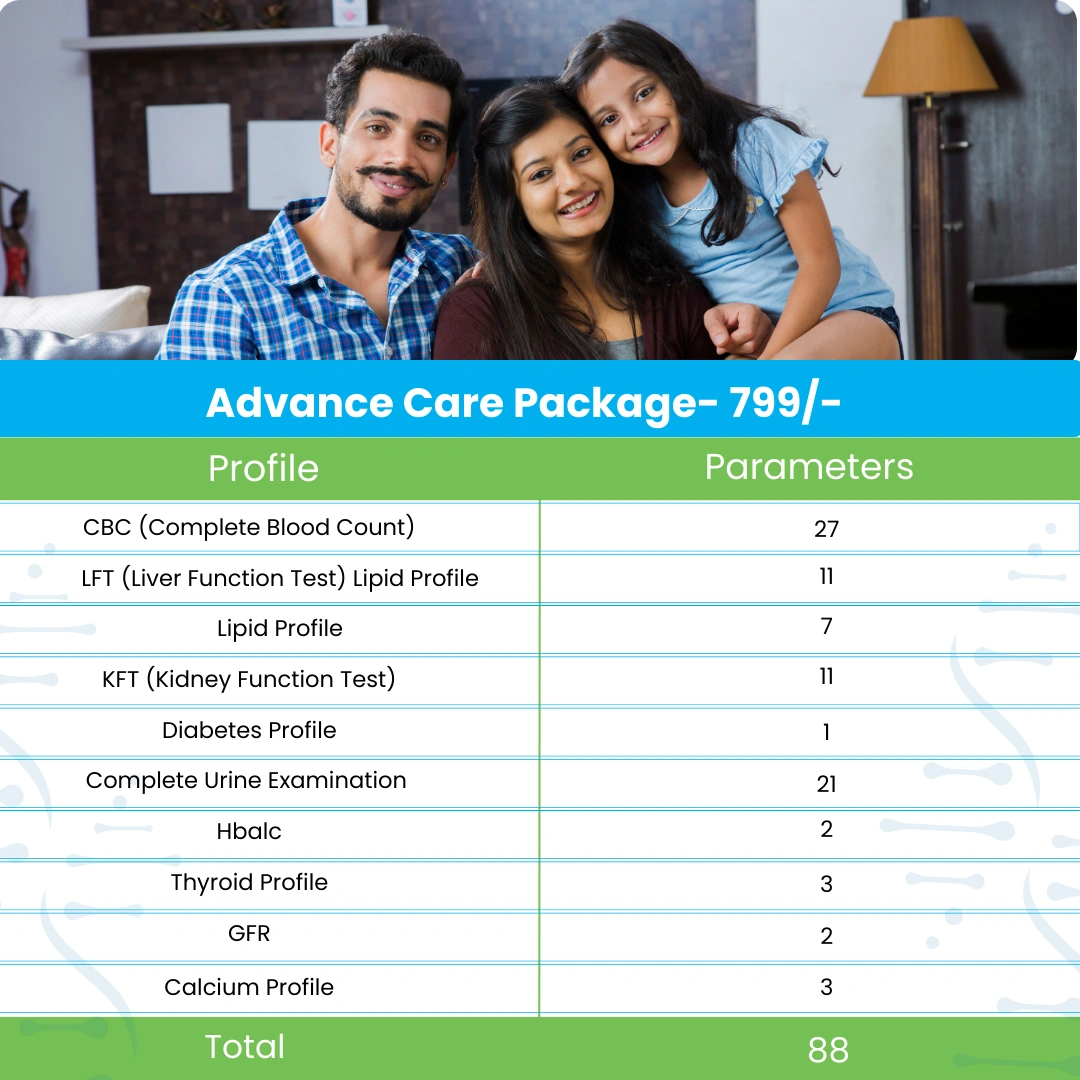














Reviews
There are no reviews yet.